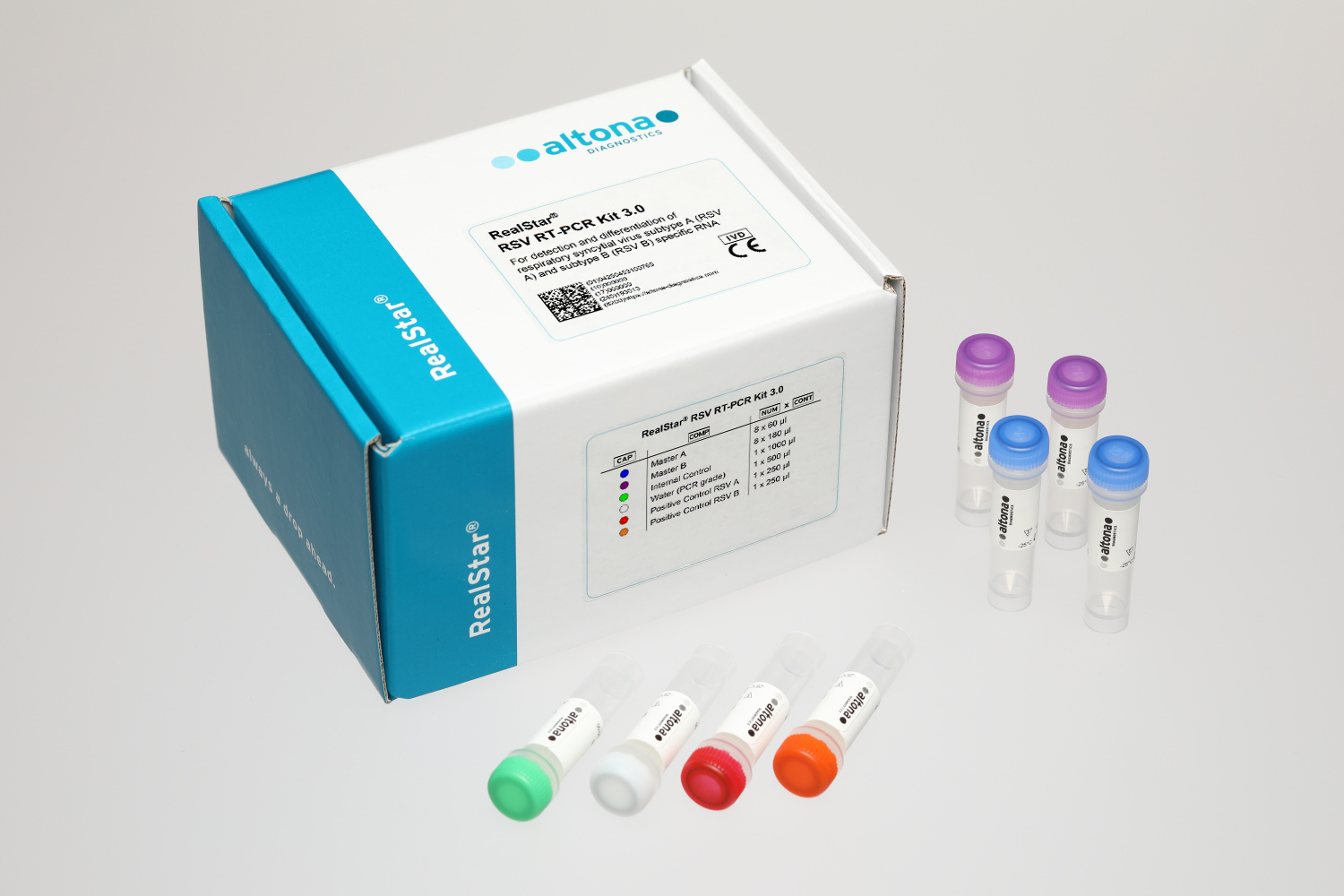
Description
- Mx 3005P™ QPCR System (Stratagene)
- VERSANT® kPCR Molecular System AD (Siemens Healthcare)
- ABI Prism® 7500 SDS (Applied Biosystems)
- ABI Prism® 7500 Fast SDS (Applied Biosystems)
- Rotor-Gene® 6000 (Corbett Research)
- Rotor-Gene® Q5/6 plex Platform (QIAGEN)
- CFX96™ Real-Time PCR Detection System (Bio-Rad)
- CFX96™ Deep Well Real-Time PCR Detection System (Bio-Rad)
- LightCycler® 480 Instrument II (Roche)