Description
Intended use
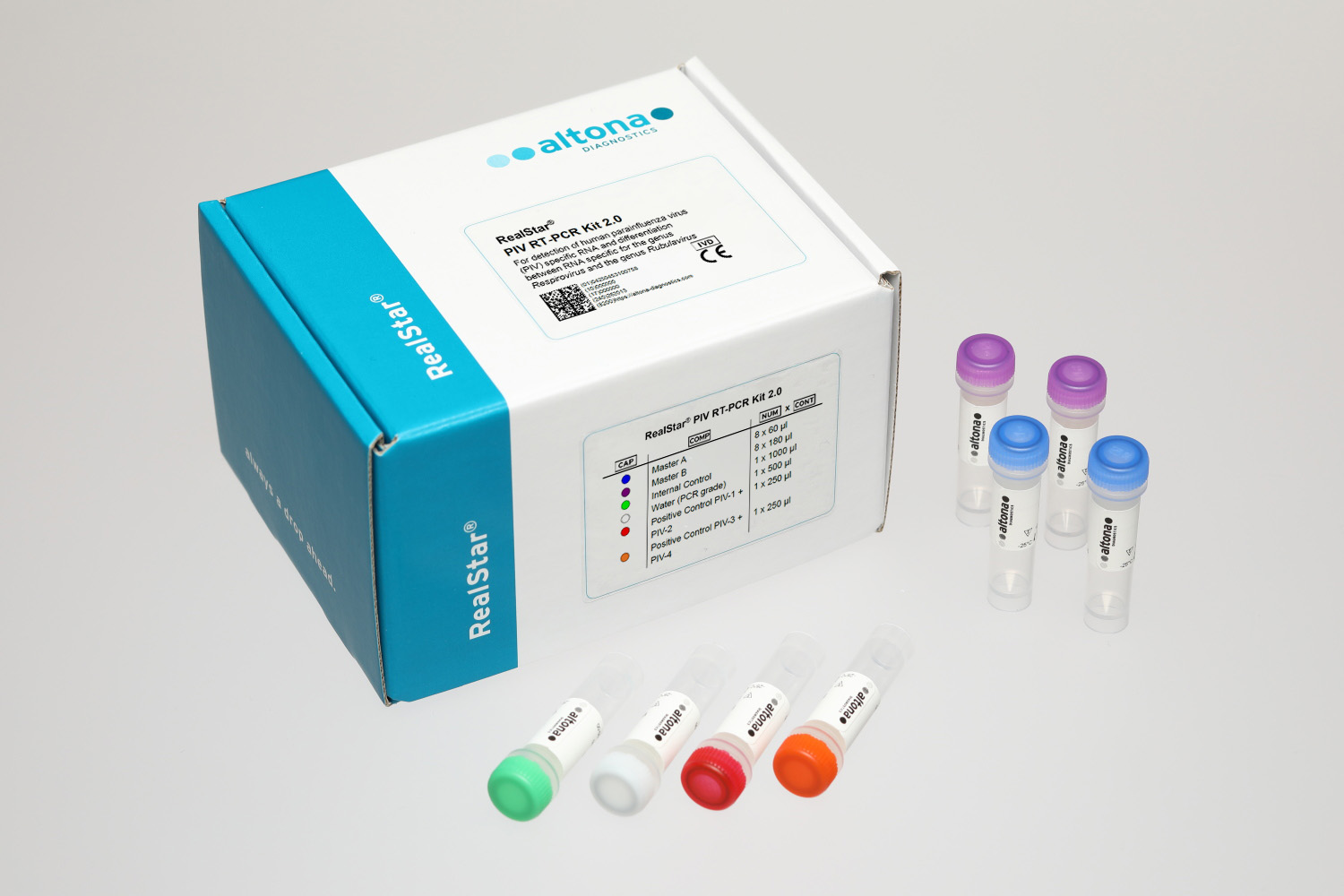
The RealStar® PIV RT-PCR Kit 2.0 is an in vitro diagnostic test, based on real-time PCR technology, for the qualitative detection of human parainfluenza virus (PIV) specific RNA of the species 1, 2, 3 and 4 (PIV-1, PIV-2, PIV-3, PIV-4). Furthermore, the test allows the differentiation between RNA specific for the genus Respirovirus (PIV-1 and PIV-3) and the genus Rubulavirus (PIV-2 and PIV-4).
Specifications
Key features
- Detection of human parainfluenza virus 1–4 specific RNA
- Differentiation of Respirovirus and Rubulavirus specific RNA
- These tests can be run in parallel: hMPV 2.0, Influenza S&T 4.0, PIV 2.0 and RSV 3.0
- Ready-to-use kit including Internal Control and Positive Control
- CE-IVD marked in vitro diagnostic test
For use with
- Mx 3005P™ QPCR System (Stratagene)
- VERSANT® kPCR Molecular System AD (Siemens Healthcare)
- ABI Prism® 7500 SDS (Applied Biosystems)
- ABI Prism® 7500 Fast SDS (Applied Biosystems)
- Rotor-Gene® 6000 (Corbett Research)
- Rotor-Gene® Q5/6 plex Platform (QIAGEN)
- CFX96™ Real-Time PCR Detection System (Bio-Rad)
- CFX96™ Deep Well Real-Time PCR Detection System (Bio-Rad)
- LightCycler® 480 Instrument II (Roche)