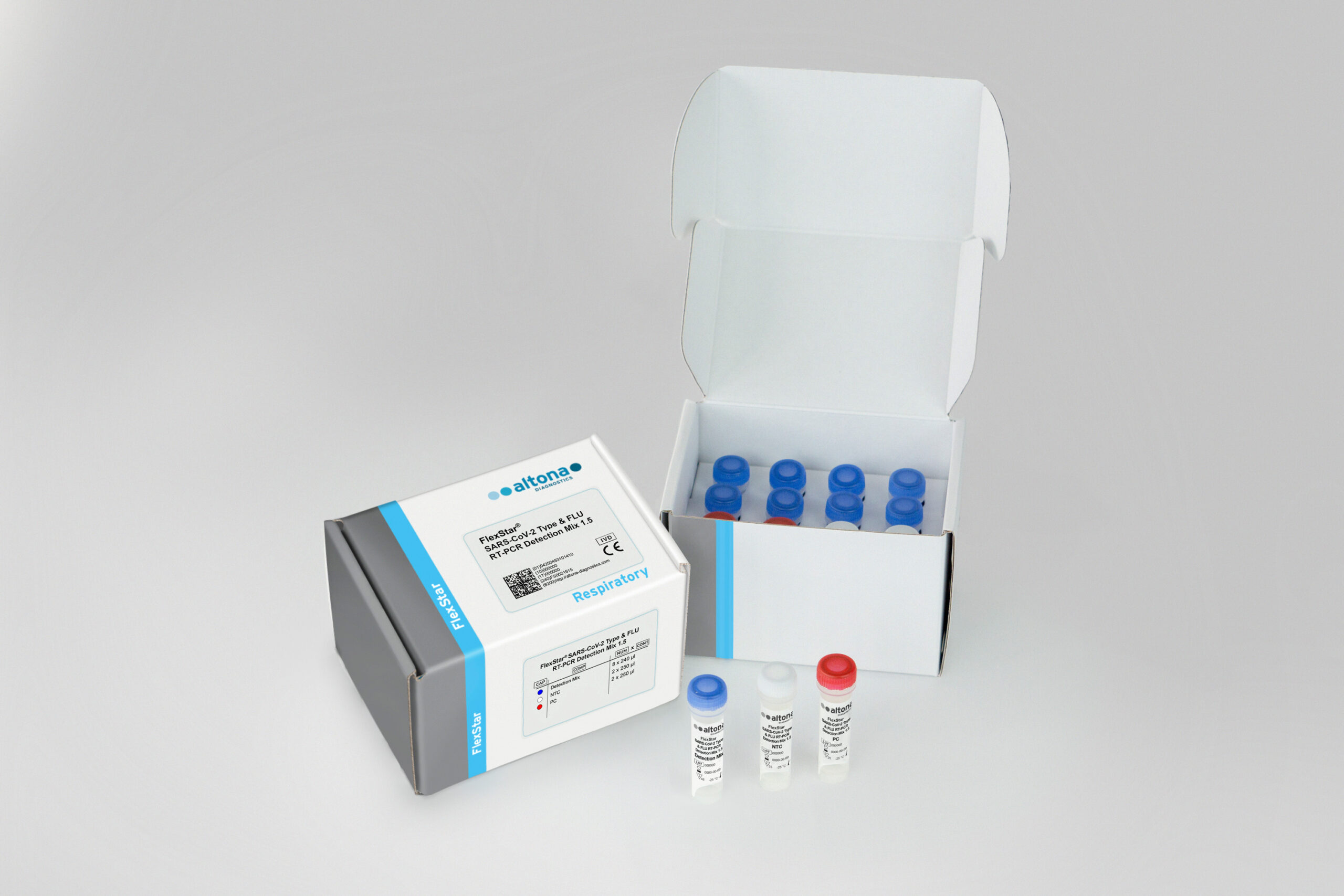
Description
Intended use
The FlexStar® SARS-CoV-2 Type & FLU RT-PCR Detection Mix 1.5 is an in vitro diagnostic test, based on real-time PCR technology, for the qualitative detection and differentiation of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and influenza virus specific RNA in human respiratory swab specimens. The SARS-CoV-2 detection is based on the parallel detection of the E gene of lineage B-beta coronavirus (including SARS-CoV-2) and the S gene of SARS-CoV-2. Validated for use with the AltoStar® Automation System AM16 for nucleic acid extraction, the AltoStar® Internal Control 1.5 and the FlexStar® (RT-)PCR Amplification Mix 1.5.
Specifications
Key features
- For detection and differentiation of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus specific RNA (E gene and S gene)
- Dual target detection SARS-CoV-2
- Parallel detection of SARS-CoV-2 virus and influenza virus A and B specific RNA in a single reaction
- CE-IVD marked in vitro diagnostic test
- Analytical sensitivity reported in IU/ml (SARS-CoV-2) and copies/ml (influenza virus A and B)
Limit of detection (LoD)
| LoD E gene | 226 IU/ml |
| LoD S gene | 201 IU/ml |
| LoD Influenza A virus | 341 copies/ml |
| LoD Influenza B virus | 432 copies/ml |
Sample types
The following sample type is validated for use with the FlexStar® SARS-CoV-2 Type & Flu RT-PCR Detetion Mix 1.5:
Human respiratory swab specimens
For use with
- LightCycler® 480 Instrument II (Roche)
- Rotor-Gene® Q5/6 plex Platform (QIAGEN)
- CFX96™ Dx System (Bio-Rad)
- CFX96™ Deep Well Dx System (Bio-Rad)